MEGA-SEL: Background and Field Testing
Background
MEGA-SEL is the result of research in the methodologies of selenium uptake that began in 1983. The objective of these studies was the development of a means of delivering selenium while controlling doses and absorption in clinical oncological research.
The typical means of introducing selenium is parenterally, thus easily controlling dosages. Site location differences apparently caused absorption differences and a means of oral delivery appeared to be an attractive alternative. Because of the chemistry of available stable selenium compounds, there were problems in the assimilation of basic selenium while constituted both as an admixture and as a compound. The acidity of typical compositions plus the high acidity of the stomach and gastrointestinal system, retarded the absorption of selenium into the bloodstream.
Administrative methodology studies were focused on a means to deliver selenium to subjects in oncological studies for use in chemo-therapy regimes. Therefore, the developers worked to establish a composition which was both extremely palatable and readily absorbed. The result was the development of an oral compound which did not impair the effectiveness of the selenium and was also extremely palatable. It was found that this selenium compound was particularly effective in inhibiting the occurrence of tumors in healthy mammals.
Demand from the equine industry for a safe and effective oral selenium and vitamin E product stimulated the application of the patented compound to horses. Many veterinarians and horse owners were aware of anecdotal accounts of immediate death after administration of injectable vitamin E / selenium preparation. (These deaths appear due to an anaphylactoid sensitivity of the horse to a carrier ingredient in the injectable preparations and not to the toxicity of selenium or vitamin E.)(1) and the relative ineffectiveness of available oral selenium supplements. Therefore, subsequent to the clinical testing, the compound was tested on equine subjects to determine its absorption, palatability and overall effectiveness. This equine selenium and vitamin E compound is trademarked MEGA-SEL.
Field Testing
Following are the results of field testing to determine the absorption of MEGA-SEL Equine Selenium and Vitamin E Compound in horses.
Baseline tests were secured prior to administration of a MEGA-SEL program. Each horse used in the test had been fed grain with feed-mill mixed selenium prior to the baseline test. Four of the 23 horses tested had been receiving additional powdered supplementation prior to administration of the MEGA-SEL program. Powdered selenium supplementation was discontinued upon addition of MEGA-SEL to diets.
Thirty days subsequent to the baseline tests, blood samples were drawn and submitted for analysis by technicians at Michigan State University’s Animal Health Diagnostic Laboratory in East Lansing, Michigan. The results are graphed herein and show excellent absorption rates for MEGA-SEL when supplemented to equine diets.
Veterinarians, owners and trainers recognized many benefits from the increase in selenium level, including: reduced muscle soreness, alleviation of tying-up syndrome, lower SGOT, greater stamina, more energy and willing attitude, improved coat and hoof condition, etc. No deleterious side effects were seen.
Field Testing Results
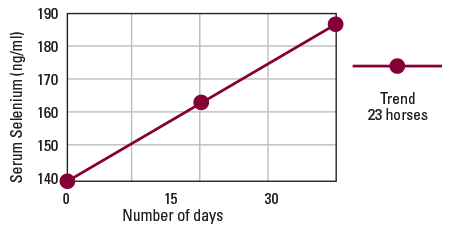
Note: The values shown on the graph represent average serum selenium levels. Individual animals may be higher or lower.
(1) “Nutrient Requirements of Horses”, National Research Council

